  Nor do two dienes combine easily upon heating to give eight-membered rings. After all, we’ve seen plenty of examples of things that don’t work two alkenes, for example, don’t combine to form four membered rings upon heating in the way that a diene and a dienophile combine to form a six-membered ring. What we haven’t really covered is why the Diels-Alder actually works. Generally the endo– is favored over the exo. in certain cases, mixtures of diastereomers ( exo– and endo- ) are obtained.the stereochemistry of the product can be reliably predicted from analyzing the stereochemistry of the diene and dienophile.three bonds form (two sigma bonds and a pi bond) and three bonds break (three pi bonds).The Diels-Alder reaction combines a diene with a dienophile to form a new six-membered ring. #MALEIC ACID SIGMA AND PI BONDS HOW TO#Using our previous posts on how to build up molecular orbitals, we’ll show how the Diels-Alder results from the constructive orbital overlap between the highest-occupied molecular orbital (HOMO) of the diene with the lowest-unoccupied molecular orbital (LUMO) of the dienophile.īut first, let’s recap where we are with the Diels-Alder so far: Today we’re going to go into the mechanism of the Diels-Alder reaction from a molecular orbital perspective. A Quick Recap Of The Diels-Alder Reaction Summary – The Key Role Of Orbital Symmetry In Concerted Reactions Of Pi-Systemsġ.Under “Photochemical” Conditions, The Actually Works Pretty Well.Molecular Orbitals In The Diels-Alder Reaction: Interaction of the Diene HOMO with the Dienophile LUMO Is Favorable At Both Bond-Forming Sites.Molecular Orbitals In The Cycloaddition Between Ethene And Ethene Show Why The Reaction Is Unfavorable Under “Thermal” Conditions.Concerted Reactions: When Two Bonds Form At The Same Time, Multiple Orbitals Must Overlap.Bond Formation Requires Overlap Between The HOMO Of One Molecule (The Nucleophile) With The LUMO Of Another Molecule (The Electrophile).A Quick Recap Of The Diels-Alder Reaction.This type of overlap is not possible in a reaction between two alkenes.By analyzing the molecular orbitals of the diene and dienophile in the Diels-Alder reaction, it becomes clear that constructive overlap is possible between the highest-occupied molecular orbital (HOMO) of the diene with the lowest-unoccupied molecular orbital (LUMO) of the dienophile.In order for the new bonds to form, there has to be constructive overlap between these orbitals – that is, they have to have the same phase.The new C-C sigma bonds are formed from the overlap of the pi molecular orbitals at C1 and C4 of the diene and C1 and C2 of the dienophile.The Diels-Alder reaction is a concerted reaction between a diene (with 4 pi-electrons) and a dienophile (2 pi-electrons) that forms a new 6-membered ring. 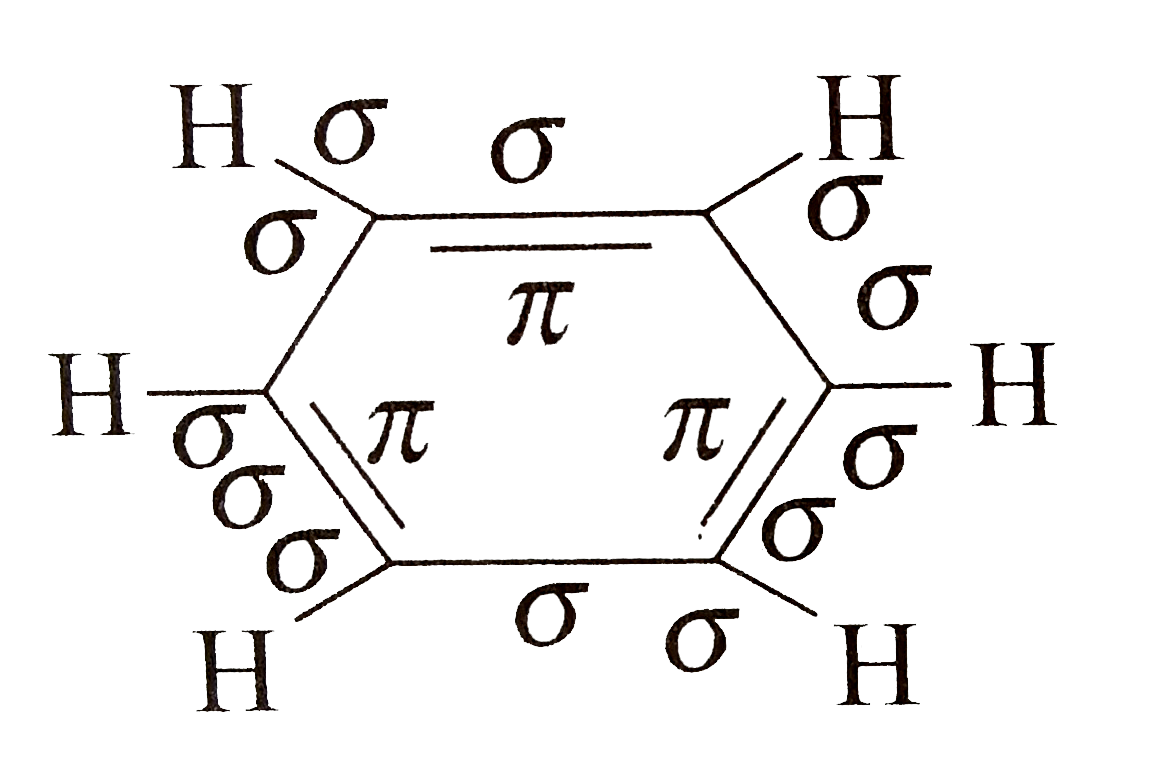
The HOMO and LUMO In The Diels Alder Reaction 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
